EFPIA seeks vendors for EU medicine verification system
Phil Taylor, 06-Apr-2012
 The European Federation of
Pharmaceutical Industries & Associations (EFPIA) has laid out
its vision of how the system of safety features enshrined in the EU
Falsified Medicines Directive (FMD) might work in a call-for-tender
document.
The European Federation of
Pharmaceutical Industries & Associations (EFPIA) has laid out
its vision of how the system of safety features enshrined in the EU
Falsified Medicines Directive (FMD) might work in a call-for-tender
document.
Unsurprisingly, the
document sticks close to the model tested in a pilot scheme in
Sweden in 2009 by EFPIA and its partners, which relied on placing
2D datamatrix codes bearing GS1-compliant codes onto packs, and
verifying them in the pharmacy as they were being dispensed to
patients.
The codes will include a randomised serial number, product number,
batch number and expiry date, and scanning should allow pharmacists
to identify fake medicines as well as genuine material that is
recalled, expired or should not be dispensed for some other
reason.
EFPIA's 'end-to-end' model - dubbed the European Medicines
Verification System (EMVS) - differs from the electronic pedigree
system envisaged under Californian law, and also from the
RxTec system of lot-level traceability and item-level
serialisation proposed by the Pharmaceutical Distribution Society
Alliance last month.
The document also describes the data management infrastructure
around the EMVS, including a series of national data repositories,
linked via an EU hub, that will "serve as the verification
platforms which pharmacies and other registered parties can use to
check a product's authenticity" and will have to handle up to 10
billion individual pack entries per year.
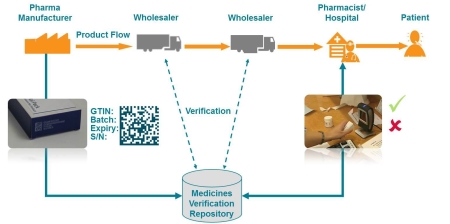
The EMVS will be able to handle parallel distribution of medicines,
as well as multi-country recalls, and a blueprint for the national
system architecture will be made available for countries that do
not currently have a national system in place at the moment.
Details of the tender are published in a request for information
document which describes the main functionality of the system and
includes a questionnaire for potential vendor companies. An
information meeting for prospective providers is scheduled to be
held on April 19 in Brussels, Belgium.
EFPIA's call for tender pre-empts the EU Delegated Acts process,
which is intended to fill in some of the key details left out of
the FMD, such as the precise nature of the 'safety features' that
must be added to prescription drug packaging.
Those involved in the Delegated Acts will be paying close attention
to the proposals, however, as EFPIA has successfully enlisted the
aid and support of the other main stakeholders in the European
pharmaceutical supply chain, including groups representing
pharmacists (the PGEU), wholesalers (GIRP) and parallel
distributors (EAEPC).
Other models vying for attention as European lawmakers deliberate
over the detail of the FMD include the eTACT
pilot developed by the European Directorate for the Quality of
Medicines & Healthcare (EDQM) and Germany's securPharm
stakeholder initiative.

©
SecuringIndustry.com




 The European Federation of
Pharmaceutical Industries & Associations (EFPIA) has laid out
its vision of how the system of safety features enshrined in the EU
Falsified Medicines Directive (FMD) might work in a call-for-tender
document.
The European Federation of
Pharmaceutical Industries & Associations (EFPIA) has laid out
its vision of how the system of safety features enshrined in the EU
Falsified Medicines Directive (FMD) might work in a call-for-tender
document.
